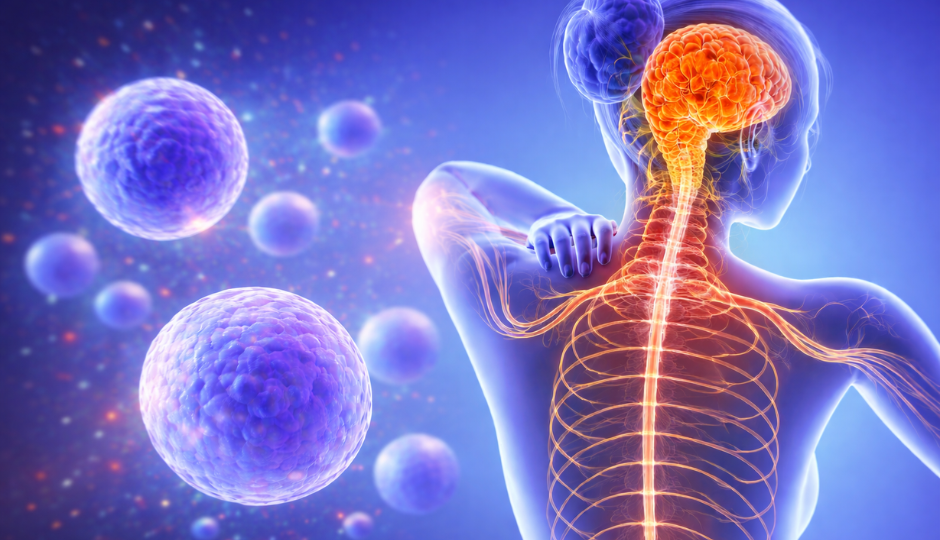
Multiple sclerosis is a chronic autoimmune condition of the central nervous system. The immune system attacks myelin, the protective sheath surrounding nerve fibres, disrupting the signals that travel between the brain, spinal cord, and the rest of the body. Over time, repeated episodes of inflammation cause demyelination and, in many patients, progressive axonal damage that leads to irreversible neurological deficit.
MS presents differently in different people. The most common form, relapsing-remitting MS (RRMS), involves episodes of worsening symptoms followed by partial or complete recovery. Secondary progressive MS (SPMS) develops in a significant proportion of RRMS patients over time, bringing steadily worsening disability without clear relapses. Primary progressive MS (PPMS) involves gradual deterioration from the outset, without distinct relapse episodes.
Common long-term consequences of MS include:
- Mobility difficulties; weakness, spasticity, and balance problems affecting walking and daily function
- Fatigue; one of the most disabling and least visible symptoms, affecting the majority of patients
- Cognitive impairment; problems with memory, processing speed, concentration, and word-finding
- Visual disturbances; including optic neuritis, blurred vision, and double vision
- Bladder and bowel dysfunction; urgency, frequency, and incontinence
- Chronic pain and sensory symptoms; numbness, tingling, burning sensations, and neuropathic pain
- Speech and swallowing difficulties; in more advanced disease
- Emotional and psychological impact; depression and anxiety are significantly more common in people with MS than in the general population
These challenges compound over time. Managing MS is not just about treating relapses, it is about preserving function, maintaining quality of life, and slowing the accumulation of disability over years and decades.