Photodynamic therapy is built around three components working together: a photosensitising agent (photosensitiser), light of a specific wavelength, and oxygen in the target tissue. None of these three elements alone causes the therapeutic effect; it is their combination that produces the anti-cancer action.
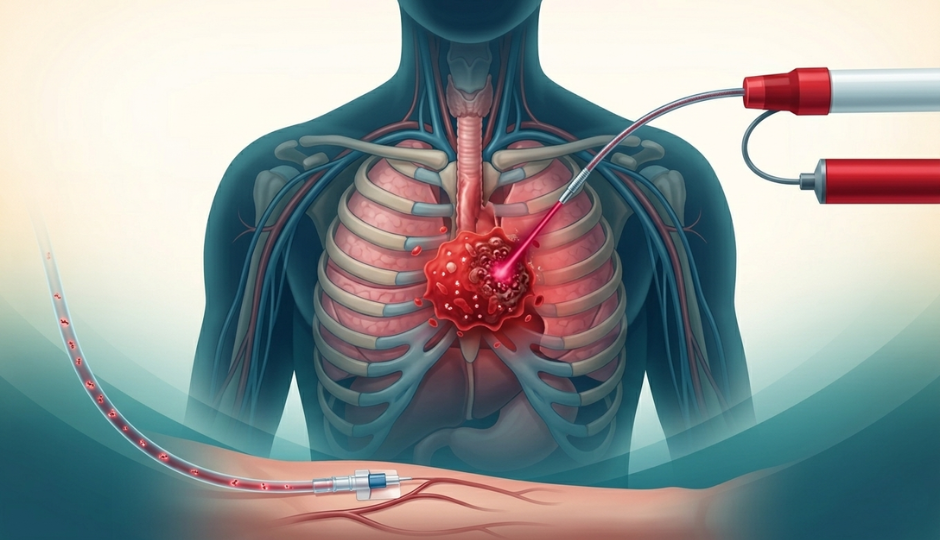
The process works as follows. A photosensitiser is administered, typically intravenously or, in some applications, topically, and allowed to accumulate preferentially in tumour tissue. Photosensitisers have a natural affinity for cancer cells and accumulate at significantly higher concentrations within them compared to surrounding healthy tissue. After a defined interval, the treatment site is exposed to light of the correct wavelength to activate the photosensitiser. Upon activation, the photosensitiser reacts with tissue oxygen to produce reactive oxygen species in particular, singlet oxygen, which are directly toxic to tumour cells.
The biological effects extend beyond the direct killing of cancer cells. PDT also damages the blood vessels that supply the tumour, cutting off its oxygen and nutrient supply and accelerating tumour death. Additionally, a body of clinical and preclinical evidence has documented that PDT can stimulate a local immune response against the tumour, potentially contributing to longer-term tumour control.
A 2024 review published in MedComm described PDT as "a temporally and spatially precisely controllable, noninvasive, and potentially highly efficient method of phototherapy", a characterisation that reflects both the approach's strengths and the precision with which it can be applied at the tumour site.