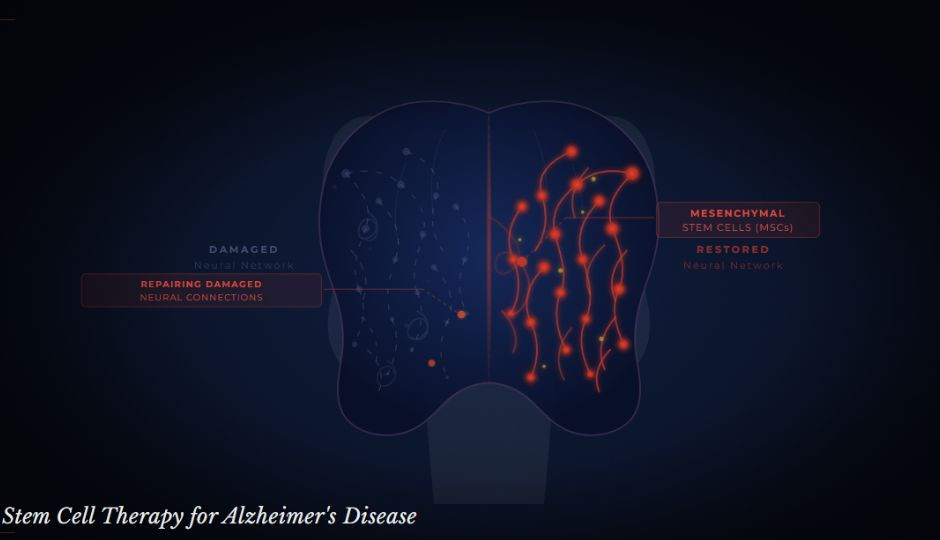
Alzheimer's is the most common form of dementia, accounting for an estimated 60–70% of dementia cases worldwide. It is a progressive neurodegenerative condition in which abnormal protein deposits, amyloid plaques, and tau tangles accumulate in the brain, disrupting communication between nerve cells and eventually causing cell death.
The disease typically progresses through three stages, though the pace varies considerably between individuals:
Early stage: Mild memory lapses, difficulty finding words, subtle changes in mood or behaviour. Many people remain independent at this stage.
Middle stage: More pronounced memory loss affecting daily tasks, confusion about time and place, changes in personality, and increasing difficulty with activities such as cooking, driving, or managing finances.
Late stage: Significant loss of independence, difficulty with basic functions including swallowing and mobility, and near-total reliance on others for care.
Long-term consequences of Alzheimer's disease include:
- Progressive memory impairment affecting both short- and long-term recall
- Language difficulties, including word-finding problems and reduced speech
- Disorientation to time, place, and eventually familiar people
- Impaired judgement and executive function
- Behavioural changes, including agitation, anxiety, and depression
- Loss of the ability to perform activities of daily living independently
- In late stages, physical decline, including difficulties with swallowing and mobility
For families, the impact extends far beyond the patient. Caring for someone with Alzheimer's is one of the most demanding experiences a person can face, emotionally, physically, and financially.